UVA Health Among First In U.S. to Implant New ICD Lead
UVA Health continues to lead the way in cardiovascular care by becoming one of the first health systems in the country to use a new type of implanted defibrillator (ICD) lead. This new lead is designed to be smaller, stronger, and more durable than previous versions. Because the lead is smaller and takes up less space within the blood vessels, it may reduce complications and improve patient comfort over time.
The procedure to implant this new type of lead, called OmniaSecure™, only requires a two-inch incision near the shoulder to reach the blood vessel.
Pamela Mason, MD, UVA Health’s chief of cardiac electrophysiology, notes that this technology is a major step forward for patients who need long-term heart monitoring, especially younger patients.
In this Q&A, Mason explores how this innovation reflects a commitment to patient-centered treatment and better outcomes.
In January 2026, UVA Health was among the first institutions in North America to implant a new defibrillator lead. What differentiates this lead from traditional designs?
One of the things that’s most important about all these devices is we want reliability and durability for our patients. Unfortunately, we have some very young patients who are having to receive these devices. And, while traditional defibrillator leads—some of them—can last for 20 years or more, if you look at the data, starting at around the 10-year mark, there is somewhat of a failure rate that becomes progressive over time.
So, if we have a patient who is 30 years old who receives one of these devices, they, at some point, are very likely to need replacement of those wires. This can be a big deal because the wires are implanted through the blood vessel and into the heart.
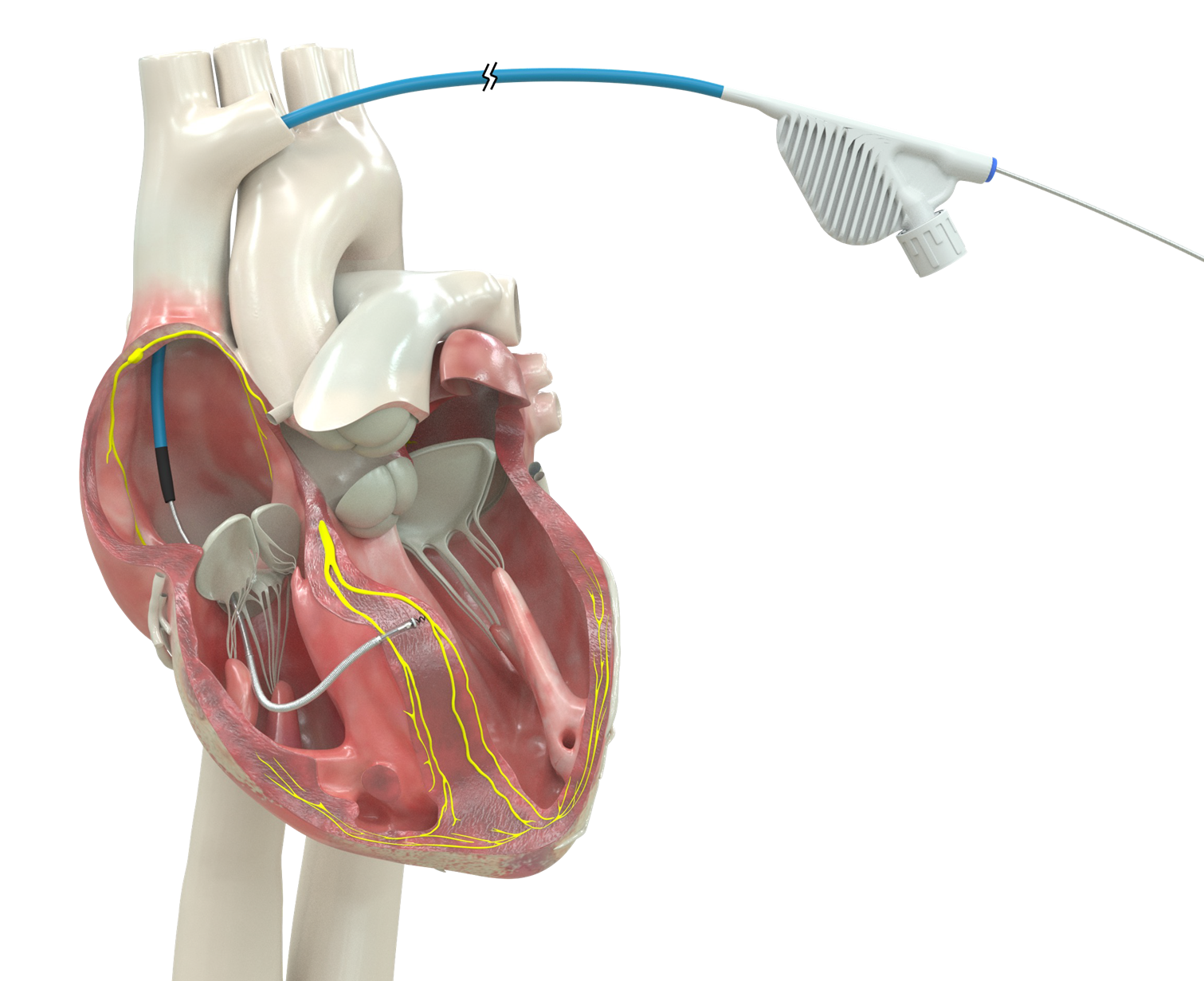
This new defibrillator lead has several potential advantages. First, it’s smaller. It’s about half the size of usual defibrillator leads, so it’s taking up less space in the blood vessel and heart. Additionally, it’s also solid construction (instead of hollow), and so we believe it’s going to be more durable. And indeed, the evidence that we have so far from modeling shows that it’s very likely to be more durable than the existing defibrillator leads for our patients.
Beyond size and durability, what additional benefits does this lead offer for patients over the long term?
Because it’s smaller and solid construction, as opposed to hollow, if we do need to take it out in the future for some reason, we believe that it’s going to be easier and more durable to remove.
Also, with the lead taking up less space, we think there’s going to be less likelihood for narrowing in the blood vessel or obstruction in the blood vessel over time.
What materials are used in the construction of this lead, and how do they impact performance and biocompatibility?
These defibrillator leads are essentially like any other electrical wire. There are interior components that are metal, which are the conductors. They’re surrounded by insulation. The insulation is often made of silicone, or it can be made of polyurethane. Many of the interior components are titanium.
We avoid nickel in these products because we don’t want to put anybody at risk for allergies.
Which patient populations are best suited for this lead, and how do you think about patient selection?
I think one question is, ‘Is there anybody who isn’t better suited for this lead, if it does have these advantages?’
But again, we really are thinking about this product for our very young patients. We really want to make sure that they have a good working device for as long as possible.
What has been your early clinical experience with this lead at UVA Health?
Here at UVA Health, we were actually part of the original clinical trial. So, prior to this device even coming to market, we had already implanted about 50 of these leads as part of the clinical trial.
Since the lead came to market, we were included in the earlier release, which is part of the reason why we were one of the first institutions in the country to implant this clinically. So far, we’ve implanted probably around 10 of those.
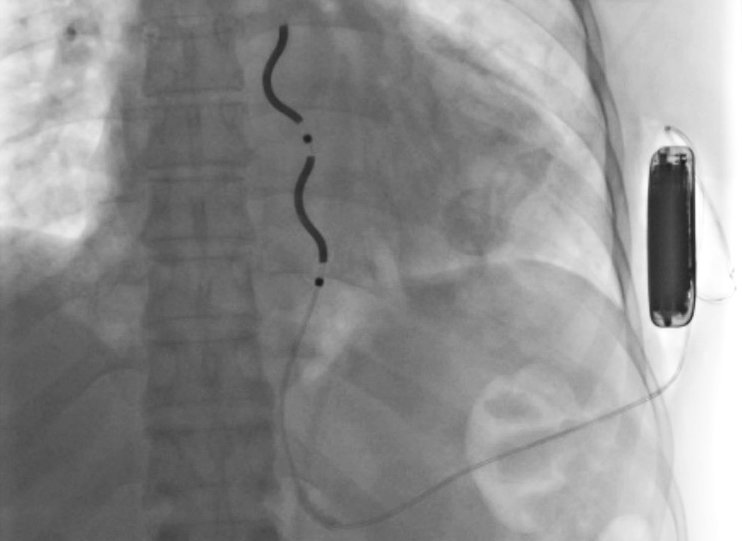
Innovating Arrhythmia Care
UVA Health was also among the first to implant the extravascular implantable cardioverter-defibrillator (EV-ICD), an ICD with leads outside of the blood vessels.
For referring providers who want to learn more or consider this technology for their patients, what’s the best way to connect with your electrophysiology team at UVA Health?
We’re obviously happy to see patients at any time in clinic to discuss. We have comprehensive discussions with patients. We have a lot of different products available. There are advantages and disadvantages of all of them.
We have the full gamut of different device options for patients, and we really do comprehensive discussions and reviews of those different options with patients so they can make informed decisions. We’re really proud of the team we have at UVA Health, and we’re really proud of what we can offer.